One-Liner: This project will design a new RNA therapeutic that nudges the longevity-linked FOXO3 gene into its health-promoting form to halt the tissue breakdown behind degenerative disc disease—opening a precise, scalable route to drugs that extend healthspan.
The proposal is published here for comments, before putting it up for vote on Snapshot.
Revision Notes
-
This proposal is a revised version of VDP-146, which was approved on Discourse but did not reach quorum in the on-chain vote. The revision incorporates community feedback and introduces additional improvements.
-
Significant changes:
-
Prioritizes a go-to-market indication from the outset
-
Includes an intermediate go / no go milestone, capping the initial risk at half of the full proposal budget
-
Gives ownership of developed IP to VitaDAO (rather than licencing)
-
Plans an IPT raise with $50k anchor funding by VitaDAO
-
Includes an expression of interest by SpineDAO to contribute funding and expertise
-
Increases the budget by $50k to advance the asset further
-
Removes c. elegans testing
-
Longevity Dealflow Team
-
Senior Reviewers: the review from VDP-146 remains largely applicable, since this proposal builds on and refines that version.
-
Shepherd: Paolo Binetti
-
Squad Members: Rhys Anderson, Daniel Hussey, Matt McAlister
-
Sourced By: Adrian Matysek
Project Team
-
Principal Investigator: Lorna Harries, Ph.D.
-
VitaDAO Project Lead: TBD
Simple Summary
Dr. Lorna Harries’ lab has identified an innovative way to modulate FOXO3, a well known and yet undrugged longevity target, and propose leveraging it to address chronic lower back pain stemming from degenerative disc disease. They plan to make short, lab-made modified RNA molecules (antisense oligonucleotides) that selectively switch FOXO3’s “good” message on, first in human cells and mini-disc models. Success would yield 1) a lead drug candidate, 2) proof it can restore disc health, and 3) the data needed for patenting and further safety and efficacy testing in animal models (not included in this project). Because this method is highly specific and can be injected straight into the spine, it promises fewer side effects than current steroids or surgery. The same molecules could later be adapted to treat many other age-related conditions, ultimately helping people stay healthy for longer.
Problem
FOXO3, a key yet undrugged longevity gene
To influence multiple facets of ageing simultaneously, we must identify ‘hub’ genes that coordinate multiple cellular processes. The FOXO3 gene is one of four human FOXO genes that forms part of an evolutionarily conserved central network orchestrating autophagy, mitochondrial quality control, DNA-damage repair, metabolic flexibility, and inflammatory restraint—integrating signals from multiple pathways (2-5). As such, it has been implicated in the regulation of ageing (3) and its associated diseases (6-8). One haplotype of FOXO3 (rs13217795-rs4946932-rs9400239) is reproducibly associated with a ≈36% increased chance of exceptional longevity across >11 human populations (2). Despite this compelling genetic validation, FOXO3 has never been successfully pharmacologically targeted. There have been previous efforts to increase FOXO3 activity using agonists, and benefits have been demonstrated in cells, tissues, and human participants (9, 10), but the molecules concerned were not specific to FOXO3 and had pleiotropic effects on a variety of crucial cellular signalling pathways intersecting with FOXO3 (and the other FOXOs) such as AKT, PI3K and AMPK.
For more background information on FOXO3, please see the “Problem” section of VDP-146.
Lower back pain, a serious unmet need
Lower back pain is the leading cause of physical disability, and Degenerative Disc Disease (DDD), also known as Intervertebral Disc Degeneration (IDD), is the most common cause of lower back pain; 12% to 16% of all adults visit their GP every year for this reason. The UK Office for National Statistics estimates that the annual incident population experiencing back pain is about 3.5 million and the prevalent population experiencing back pain is 17.3 million, with 3.1 million adults suffering during the entire year in the UK alone. Over a 1-year period, 116 million production days were lost to back pain.
There are currently few treatment options. The standard of care (SoC) for DDD is steroids (which have many adverse side effects and adverse long term outcomes) or surgery—which involves protracted recovery time, infection risk (and associated risk of post-operative delirium), and anaesthesia-induced mortality in older patients. Surgery is invasive, costly, not durable, and can even worsen the disease: for example arthrodesis of two vertebrae, induces additional stresses on nearby intervertebral discs (IVD), accelerating their degeneration, leading to the need for more procedures. In summary there is currently no good option, and none disease-modifying.
Solution
Drugging FOXO3 by modulating its isoforms
Prof. Harries’ team discovered that the association between extreme lifespan and FOXO3 genetic variation was likely mediated by the production of alternatively expressed mRNA isoforms of FOXO3 (11).
FOXO3 expresses three linear mRNA isoforms and one circular RNA. The full-length FOXO3-long isoform encodes the complete fork-head DNA-binding domain and is ubiquitous, whereas FOXO3-short lacks key regulatory motifs and is enriched in ageing muscle and degenerating tissues (11). Ageing and metabolic stress progressively tilt the balance toward the short form, diminishing the cell’s ability to mount stress responses and maintain tissue integrity (17). Restoring the youthful long-to-short ratio may therefore represents a precise, mechanistically grounded entry point for gerotherapeutic intervention.
For additional information on FOXO3 isoforms, please see the “Solution” section of VDP-146.
Oligonucleotide modality
The team proposes oligonucleotide drugs designed to allow regulated control of patterns of FOXO3 isoform expression. They can enhance gene expression with small activating RNAs (saRNAs) or by steric hindrance of negative regulatory elements. Conversely, genes can be repressed by RNAi or steric blockage of sequences necessary for expression. Steric hindrance ASOs have already proven to be safe and effective in the clinic (for example in SMA and DMD).
This approach is based on the PI’s almost 30 years’ experience in RNA biology and knowhow in oligonucleotide design and delivery developed in her capacity as founder and CSO of SENISCA Ltd. (www.senisca.com). Such an intervention would have an extremely safe usage profile compared with a small molecule; oligo drugs act on a specific region of 20-25 nucleotides at the site of action on the target gene alone and bind nowhere else if correctly designed (22). They are the fastest growing drug modality presently with 4 new approvals in 2023 alone. Depending on the target organ, they can be delivered using lipid nanoparticles, or even naked in saline if locally (22). They can be signposted to specific organs and tissues using peptides, antibody conjugates, or hydrogel carriers (23) and allow researchers to drug ‘undruggable’ targets such as transcription factors, which are notoriously hard to moderate with small molecules. Although local delivery is now fairly routine, systemic delivery of oligonucleotides has historically been a challenge, but new technologies for delivery to specific body compartments are under rapid development (24).
FOXO3 for DDD: a disease modifying approach
FOXO3 expression is strongly correlated with DDD in both mouse and human (18), and has been associated with extracellular matrix degradation and nucleus pulposus cell death both in vitro and in vivo (19). Furthermore, FOXO3 overexpression ameliorates intervertebral disc degeneration by decreasing NLRP3-mediated pyroptosis (a form of cell death) (20).The balance of FOXO3 isoforms is known to be important in bone remodelling. Absence of FOXO3 in mice leads to elevated numbers of osteoclasts, responsible for bone resorption. Importantly, the long and short isoforms of FOXO3 play unique roles, with short isoforms in particular suppressing bone remodelling (21). Interventions that are capable of differentially affecting individual FOXO3 isoforms may therefore prove a promising option for degenerative conditions of the bones.
The therapeutics proposed by the lab would be delivered directly to the intervertebral space by injection, a standard route administration. Oligonucleotide therapeutics have a long duration of action; in some cases up to 6 months. They would be delivered naked: the PI, through her role as founder and CSO of SENISCA, has a proven track record of delivering oligonucleotides to human primary cell models by gymnosis, and delivering nucleic acids in simple invertebrate systems is routine. Efficacy could be enhanced with a DDD-specific formulation, adding later growth potential.
Opportunity
While a gerodrug can treat or prevent multiple age-related disease and even extend lifespan, the project will focus on an exemplar and very common disease of ageing: degenerative disc disease (DDD), also known as intervertebral disc disease (IDD), for which no disease-modifying interventions exist. Demonstrating proof of principle in one condition then creates the opportunity to extend the drug’s application to additional aging-associated disorders in the future.
FOXO3 in the Context of DDD: Go to Market Strategy
When considering an exemplar disease to target with a new gerodrug, there needs to be a clear and coherent causal link to disease, an easy route of administration, relatively low heterogeneity in the patient population, and a market need. Degenerative Disc Disease (DDD) fulfills these requirements, making it a sensible choice for this gerodrug’s first outing:
-
published research connects FOXO3 with DDD.
-
the asset can be delivered locally, through a route of administration already familiar to patients and clinicians.
-
presentation is less heterogeneous than other candidate diseases (e.g. cancer, diabetes, and cardiovascular disease are majorly linked with lifestyle).
-
there is a large and growing market: about $35 billion in 2025, projected to reach $45 billion by 2029 with a 7.5% CAGR.
-
while the development space is competitive, it’s less crowded than indications like cancer, cardiovascular disease, or diabetes.
Although there are other assets in the pipeline (as is true for most diseases of ageing), the competitive space here is less crowded. Several drugs in the pipeline are either non-disease modifying (e.g. hydrogels), involve non-specific targeting of major cellular pathways which may risk tolerability or off target effects (e.g. TGFb targeting), or are biological agents such as stem cell therapies, with limited patient availability, high cost, and potential rejection issues. In contrast, a targeted AON drug has a very precise MoA, very limited off target profiles, and improved safety, particularly when administered locally, due to limited systemic exposure. Thus, an injectable, disease-modifying RNA therapy would compete favorably against implants, stem-cell products and non-specific growth-factor blockers (12).
FOXO3 in the Context of Age-Related Diseases: future label expansion
As one would expect from its pivotal position in cellular health, FOXO3 dysregulation is associated with several age-associated diseases. All of these diseases are major contributors to morbidity and mortality. The table below is an overview of the pros and cons of each opportunity, including DDD for reference.
| Disease area | Key advantages (pros) | Key challenges (cons) | Est. 2025 US market size | FOXO3 ref. |
|---|---|---|---|---|
| Degenerative disc disease (DDD / IDD) | • Local intradiscal injection avoids systemic PK issues • Moderately crowded pipeline (≈15 active assets vs. >100 in CVD) • Disc height & pain endpoints well-validated |
• Chronic low-back-pain space still competitive (cell, hydrogel, peptide trials) | $35 B global spine-pain therapeutics & surgery | (12) |
| Cardiovascular disease (CVD) | • Huge market • LDL-C, BP & troponin are hard biomarkers • Clear FDA pathways & huge payer interest |
• Highly crowded (>100 late-phase drugs) • Requires systemic delivery • Lifestyle & poly-morbid confounders |
$160 B drugs only | (13) |
| Neuro-degenerative disorders | • Orphan sub-indications (ALS, HD) give regulatory incentives • Enormous unmet need; disease-modifying space still thin |
• Blood-brain-barrier delivery • High trial-failure rate • Heterogeneous patient population |
$20 B | (14) |
| Cancer | • Largest single therapeutic market • Many fast-track / breakthrough pathways • Biomarker-defined niches allow premium pricing |
• Extremely crowded (immuno-oncology, cell & gene, radioligands) • Heterogeneous patient population • Complex safety expectations |
$200 B | (15) |
| Auto-immune / immune dysfunction | • Chronic, high-value biologic segment • Clear clinical scales (DAS-28, PASI) • Potential for targeted local dosing in joints/skin |
• Humira-class biologics dominate • Heterogeneous patient population • Long systemic exposure • Infection risk |
US $170 B | (16) |
| Sarcopenia / frailty | • No FDA-approved drugs yet: first-in-class potential • Ageing biology directly relevant |
• Regulatory endpoints still debated (SPPB, gait speed) • Smaller current spend • Requires systemic delivery |
$4 B projected | (7) |
| Diabetes & complications | • Fast-growing market • Hard biomarkers (HbA1c, CGM), short PoC trials • Clear reimbursement pathways and guideline adoption |
• Very crowded (GLP-1, SGLT2, Tirzepatide class boom) • Requires chronic systemic dosing • Lifestyle & obesity confounders |
$40 B | (17) |
Relevance to Longevity
By precisely re-balancing the long- and short-isoforms of the longevity gene FOXO3, this project targets a central node that orchestrates autophagy, mitochondrial maintenance, DNA-damage repair, and anti-inflammatory signaling—processes that collectively slow the functional decline of multiple tissues. The proof-of-concept focus on degenerative disc disease (DDD) offers a clear clinical path, yet the same antisense strategy can later be retargeted systemically or to other organs to counteract sarcopenia, vascular aging, immune dysfunction, and metabolic disease. Because the intervention modulates an evolutionarily conserved hub rather than a single downstream pathology, it holds the potential to extend healthspan—the period of life spent free from chronic disability—as well as lifespan.
IP Roadmap
The output from phase 1 of this work would be prototype oligonucleotides that can bring about the required changes to FOXO3 isoform usage, and ultimately FOXO3 activity, and a demonstration that treatment with one or more of these assets is able to bring about an attenuation of senescence and disease markers in 2D and 3D tissue culture systems. This would provide enough data to make an initial patent filing. Following initial filing, claims would be bolstered by provision of additional data from in vivo model systems within the following 12 months, but this would be contingent on further funding.
Experimental Plan
Hypothesis: Targeted moderation of FOXO3 isoform expression profiles will attenuate cellular ageing and tissue health parameters in vitro and ex vivo and will bring functional benefits in vivo.
Phase 1: IP Building Round (to be covered by the current fundraise)
Milestone 1a: AON Design and Validation in Monolayer Culture
Objective: To design and validate antisense oligonucleotides (AONs) that effectively and specifically modulate FOXO3 isoform expression in human monolayer culture of primary fibroblasts (NHDF), Human Nucleus Pulposus Cells (HNPC) and Human Annulus Fibrosus Cells (HAFC).
Workplan: The Harries’ lab will design and validate antisense oligonucleotides (AONs) attenuate FOXO3 isoform usage by the use of steric hindrance translation blockers against the isoform-specific translation start sites of FOXO3-short or -long isoforms, or by blocking negative regulatory elements specific to each isoform to manipulate FOXO3 isoform expression patterns. These are standard technologies which are routinely used in both the Harries’ academic team and her industrial team at SENISCA (we have several oligonucleotides using a similar MoA). The efficiency of isoform switching will be measured by band size difference and band intensities by Western blot using an anti-FOXO3 antibody that binds an epitope present in both isoforms as well as by RT-qPCR. By these means, it is possible to produce cells expressing primarily FOXO3-long, primarily FOXO3-short, or both. This will demonstrate the correct molecular activity of the new AONs, as well as providing early target engagement data and a tool for high throughput screening of chemistries and length changes in phase 2.
Outcome: Identification of at least two lead AON candidates that demonstrate potent and specific modulation of FOXO3 isoforms and a reduction in senescence markers. This will provide the foundational evidence that our asset is producing the correct molecular outcome.
Milestone 1b: Evaluate AON Efficacy in 2D Culture
Objective: To determine if the changes in FOXO3 isoform expression translate to a functional rescue of the senescent phenotype in cells relevant to Degenerative Disc Disease. To determine if 1) AONs alter FOXO3 activity and subcellular localisation, and 2) if these changes translate to a functional rescue of DDD phenotypes, including changes in autophagy, mitochondrial function, as well as the phenotype and rate of accumulation of senescent cells.
Workplan: The subcellular localisation of proteins encoded by FOXO3-long and FOXO3-short isoforms will be assessed in primary human fibroblasts (NHDF) Human Nucleus Pulposus Cells (HNPC) and Human Annulus Fibrosus Cells (HAFC) monolayer by immunofluorescence microscopy. Cells can be induced to a degenerative state resembling DDD using TNFa and IL-1b under serum limiting conditions (27). Changes in the relative ability of FOXO3 isoforms to transactivate a commercially available luciferase reporter construct containing FOXO binding sites will then be assessed as a measure of FOXO3 activity. Effects on mitochondrial function will be assessed using live cell measurement of mitochondrial respiration and glycolysis on the Seahorse XF24 platform (20,21) following oligonucleotide manipulation. The team will also measure the impact of isoform usage on rate of senescence by monitoring senescence-associated beta galactosidase (SA-beta-Gal) activity, EdU incorporation (for effects on proliferation), gamma H2AX (for the accumulation of senescence-associated DNA damage foci), and autophagy using fluorescence microscopy on the Operetta high content imager where we have built a unique machine learning algorithm for the classification and quantification of senescent cells. They will also assess effects on the senescence-associated secretory phenotype (SASP), molecular markers of senescence (CDKN2A [p16], CDKN1A [p21], TP53 [p53] and LAMINB1) and disease-associated features (MMP9, MMP13, MMP3, ACAN) by quantitative PCR and ELISA as appropriate.
Outcome: This work will provide validation of the impact of our nascent therapeutics on the activity and subcellular localisation of FOXO3 isoforms, and on mitochondrial function, senescence kinetics, autophagy, and cell survival in human primary cell types relevant to DDD in 2D.
→ Go-No/Go Point After Milestones 1a and 1b
The decision to proceed to the next phase will be based on the successful achievement of Milestones 1a and 1b: specifically, the identification of AONs that can robustly shift FOXO3 isoform ratios and produce a statistically significant improvement in at least two key markers of cellular health (e.g., reduced senescence, increased matrix production).
Milestone 2: Evaluate AON Efficacy in a Human ex vivo Model
Objective: To evaluate the ability of AONs targeting specific FOXO3 isoforms to modulate markers of disc health and degeneration in a physiologically relevant ex vivo model of degenerative disc disease (DDD).
Workplan: Following assessment of the influence of AONs targeted to different FOXO3 isoforms in cells relevant to DDD in monolayer, I will assess the impact of these assets in ex vivo organotypic organ-on-a chip cultures (and if available, primary human IVD explants). These ex vivo platforms will incorporate human primary nucleus pulposus (NP) and annulus fibrosus (AF) cells induced into a DDD-like phenotype using TNF-α and IL-1β. (27). The organ-on-a-chip configuration enables precise spatial arrangement of NP and AF cells to replicate native disc architecture (e.g. NP cells sit in the centre of the tissue while AF cells reside around the periphery), as well as co-culture with additional cell types such as stromal fibroblasts.This setup will also allow evaluation of how treatment affects the paracrine signaling of aged or damaged cells on unconnected cell populations. Disease-relevant outcomes will be assessed, including expression of collagens, fibromodulin, IL-6, IL-8, aggrecan (ACAN), MMP13, MMP9, and MMP3, along with morphological analysis.
Outcome: This work will generate validation data demonstrating the impact of FOXO3-targeted AONs on molecular and structural features of disc degeneration in a human-relevant ex vivo system, supporting further development of these therapeutics for degenerative disc disease.
Figure 3: Diagrammatic workplan of proposed project. MILESTONE 1a is the design of appropriate oligonucleotides. MILESTONE 1b is the evaluation of their performance in an in vitro system. MILESTONE 2 is evaluation of oligos in an ex vivo human disease model system.
Go/No Go Milestones for Phase 2
-
Identification of an appropriate prototype oligo that produces the desired effect on FOXO3 isoform usage and activity in the 5nM to 400nM range.
-
Identification of an appropriate prototype oligo that has beneficial effects on cellular ageing and disease markers in 2D monocultures and 3D human primary cell organoid models of DDD.
Phase 2: Translation Round (to be covered by a subsequent fundraise)
The first steps in translating the IP arising from phase 1 would be to optimise the oligo sequence and chemistries. This is something I do routinely in my capacity as CSO for SENISCA. Briefly, the optimal oligo sequence can be deduced by ‘walking’ the oligo base by base across the sequence. Following this, the lead sequence can then be assessed as different nucleoside and backbone moieties and different dosages (typically 25nM to 400nM) firstly in vitro for target engagement.
In vivo PK/PD and Toxicology Studies (CRO)
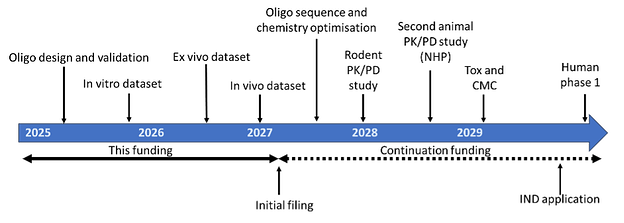
This would use an appropriate rodent animal model for DDD. There are several well used mouse models suitable for this work (lumbar spine instability models, chemonucleosis, compression). I have an established network of CROs through work undertaken by SENISCA. Outcome measures would be conventional macroscopic tests for pain and behaviour, as well as imaging and molecular features of inflammation, senescence, and matrix remodelling. Standard toxicology assessment would also be undertaken. CMC and toxicology packages (traditionally in 2 mammalian species) are undertaken somewhat later for AON drugs than they are for a conventional small molecule, as the test item has to be exactly what is proposed in the IND package. Depending on the oligo sequence and the degree of sequence conservation, the preceding PK/PD work may require the use of an oligo designed to the mouse, rather than the human sequence, but this is a conventional workflow for non-conserved oligos. Toxicology and CMC work would begin once a candidate molecule has been identified. Figure 4 below illustrates the path to IND status we would take. This is by no means a complete and granular plan but highlights some of the future work that would be necessary towards IND status and regulatory approval prior to entry into trials.
Figure 4: The path to clinical for a new oligonucleotide geroprotective drug. The initial phases of this work, funded by initial investment, would cover the production of prototype oligonucleotides that can be protected by patent, and evaluation of these in vitro, ex vivo and in vivo. Following this phase, the next steps are to optimise the oligonucleotide sequence and chemistry, and then to determine biodistribution and gross toxicology in a higher animal model system, which is usually rodents. This determines whether it is possible to deliver naked oligos in saline to the target cells and organs. Following on from this, we would pursue a traditional preclinical package consisting of CMC, safety toxicology, and PK/PD. The nature of this and the identity of the animal models involved would depend on the outcome of our earlier work, as some oligos will be conserved and can be assayed for efficacy in rodents, while others may be primate specific and would require non-human primates. Milestones and estimated timing to eventual IND approval are given.
Budget - Phase 1
Milestone 1a and 1b
| Item | UK £ | US $ |
|---|---|---|
| Staffing | ||
| PI Lorna Harries Project Management 2hr Per Week | 18371 | 24885 |
| Postdoctoral Research Associate (PDRA) to Carry Out Work | 70124 | 94989 |
| Reagents for Milestone 1a: Design and Validation of AONs | ||
| AON Oligos (8 sequences; £200 each) | 1600 | 2168 |
| General Cellular/Molecular Consumables | 2500 | 3387 |
| Oligofectamine | 1600 | 2168 |
| Primary Cells (AF and NP) | 2200 | 2981 |
| Specialist Tissue Culture Reagents | 3000 | 4064 |
| qPCR | 1000 | 1355 |
| Laptop Computer for PDRA | 2000 | 2710 |
| Reagents for Milestone 1b: Evaluation of AONs in vitro | ||
| Agilent Seahorse XF Cell Mito Stress Test Kit - 6 x 96 Well Plates | 700 | 949 |
| Antibodies | 1250 | 1694 |
| Edu Kits | 500 | 678 |
| H2Ax Kits | 600 | 813 |
| Immunofluorescence Consumables | 750 | 1016 |
| Luciferase Reporter Gene Kits (2) | 1200 | 1626 |
| ELISA | 1500 | 2032 |
| SAB Kits | 500 | 678 |
| TUNEL | 900 | 1219 |
| Western Blotting | 500 | 678 |
| qPCR | 1000 | 1355 |
| Specialist Tissue Culture Reagents | 3000 | 4064 |
| SUBTOTAL | £114795 | $155509 |
![]() Go/No-Go Point at the End of Year 1
Go/No-Go Point at the End of Year 1
Milestone 2: Evaluation of AONs Ex vivo
| Item | UK £ | US $ |
|---|---|---|
| Staffing | ||
| PI Lorna Harries Project Management 2hr Per Week | 18371 | 24885 |
| PDRA to Carry Out Work | 70124 | 94989 |
| Reagents | ||
| OOC System | 7806 | 10570 |
| Antibodies | 1250 | 1693 |
| Immunofluorescence Consumables | 750 | 1016 |
| ELISA | 1500 | 2032 |
| SAB Kits | 500 | 678 |
| LDH | 250 | 339 |
| Western Blotting | 500 | 678 |
| Specialist Tissue Culture Reagents | 4000 | 5416 |
| SUBTOTAL | £105051 | $142296 |
GRAND TOTAL: £219,846 or $297,805
Financing and VitaDAO Funding Terms
This project will be financed through the issuance of Intellectual Property Tokens (IPTs) on Bio Protocol’s platform, anchored by a $50,000 commitment from VitaDAO on the same terms as all contributors, including aligned DAOs, such as SpineDAO.
The funding terms are an agreement for contract research services with Exeter University, granting Vitality Now, the Swiss association representing VitaDAO, ownership to the intellectual property developed by the project.
Team and Research Environment
Professor Lorna Harries
Professor Lorna Harries gained her PhD from University College London in 1994. Lorna established the RNA-mediated disease mechanisms group at Exeter in 2006 and holds a personal chair in Molecular Genetics at the University of Exeter Medical School and a position as co-founder, co-director and Chief Scientific Officer at SENISCA Ltd, a spin out company founded on the Harries lab’s research. Lorna was awarded the Diabetes UK RD Lawrence rising star award in 2011, was a team member of the Queen’s Anniversary Prize for research excellence in 2006 and a direct recipient in 2019. She was awarded the Proteomass Lifetime Career Award in 2021 for her work on senescence and RNA processing. The Harries lab has interests in -omics approaches to the study of ageing and age-related disease processes in humans. She has published over 160 peer reviewed publications in her career which have accrued over 20000 citations, and she has an H index of 56. She has a proven track record of translating basic research and is an inventor on two patents to date, with 7 more in draft.
Research Team
The scientist appointed using these funds (“PDRA” for Post Doctoral Research Associate) will be exclusively focused on this project and will benefit from the wider Harries academic team which consists of 7 individuals with multidisciplinary skills ranging from primary cell biology, multi-omics, organ on a chip systems, bioinformatics, and molecular genetics. This is complemented by the co-location of the lab to the Harries R&D team of SENISCA, for whom Lorna is Founder and CSO. SENISCA has a head count of 16 people and specialises in the development of oligonucleotide drugs.
References
-
CMO. Chief Medical Officer’s Annual Report 2023 Health in an Ageing Society. 2023.
-
Willcox BJ, Donlon TA, He Q, Chen R, Grove JS, Yano K, et al. FOXO3A genotype is strongly associated with human longevity. Proc Natl Acad Sci U S A. 2008;105(37):13987-92.
-
Chang ZS, He ZM, Xia JB. FOXO3 Regulates the Progress and Development of Aging and Aging-Related Diseases. Curr Mol Med. 2023;23(10):991-1006.
-
Tseng AH, Shieh SS, Wang DL. SIRT3 deacetylates FOXO3 to protect mitochondria against oxidative damage. Free Radic Biol Med. 2013;63:222-34.
-
Huang H, Tindall DJ. Dynamic FoxO transcription factors. J Cell Sci. 2007;120(Pt 15):2479-87.
-
Alvarez-Garcia O, Matsuzaki T, Olmer M, Miyata K, Mokuda S, Sakai D, et al. FOXO are required for intervertebral disk homeostasis during aging and their deficiency promotes disk degeneration. Aging Cell. 2018;17(5):e12800.
-
Gellhaus B, Boker KO, Gsaenger M, Rodenwaldt E, Huser MA, Schilling AF, et al. FOXO3 Knockdown Mediates Decline of Myod1 and Myog Reducing Myoblast Conversion to Myotubes. Cells. 2023;12(17).
-
Jing Y, Zuo Y, Yu Y, Sun L, Yu Z, Ma S, et al. Single-nucleus profiling unveils a geroprotective role of the FOXO3 in primate skeletal muscle aging. Protein Cell. 2023;14(7):497-512.
-
Chen X, Li M, Li L, Xu S, Huang D, Ju M, et al. Trehalose, sucrose and raffinose are novel activators of autophagy in human keratinocytes through an mTOR-independent pathway. Sci Rep. 2016;6:28423.
-
Kaplon RE, Hill SD, Bispham NZ, Santos-Parker JR, Nowlan MJ, Snyder LL, et al. Oral trehalose supplementation improves resistance artery endothelial function in healthy middle-aged and older adults. Aging (Albany NY). 2016;8(6):1167-83.
-
Frankum R, Jameson TSO, Knight BA, Stephens FB, Wall BT, Donlon TA, et al. Extreme longevity variants at the FOXO3 locus may moderate FOXO3 isoform levels. Geroscience. 2021.
-
Wu Z, Zhan W, Wu L, Yu L, Xie X, Yu F, et al. The Roles of Forkhead Box O3a (FOXO3a) in Bone and Cartilage Diseases - A Narrative Review. Drug Des Devel Ther. 2025;19:1357-75.
-
Zhao Y, Liu YS. Longevity Factor FOXO3: A Key Regulator in Aging-Related Vascular Diseases. Front Cardiovasc Med. 2021;8:778674.
-
Maiese K, Chong ZZ, Shang YC. “Sly as a FOXO”: new paths with Forkhead signaling in the brain. Curr Neurovasc Res. 2007;4(4):295-302.
-
Liu Y, Ao X, Ding W, Ponnusamy M, Wu W, Hao X, et al. Critical role of FOXO3a in carcinogenesis. Mol Cancer. 2018;17(1):104.
-
Xu S, Ma Y, Chen Y, Pan F. Role of Forkhead box O3a transcription factor in autoimmune diseases. Int Immunopharmacol. 2021;92:107338.
-
Parmar UM, Jalgaonkar MP, Kansara AJ, Oza MJ. Emerging links between FOXOs and diabetic complications. Eur J Pharmacol. 2023;960:176089.
-
Alvarez-Garcia O, Matsuzaki T, Olmer M, Masuda K, Lotz MK. Age-related reduction in the expression of FOXO transcription factors and correlations with intervertebral disc degeneration. J Orthop Res. 2017;35(12):2682-91.
-
Tan Y, Wang X, Zhang Y, Dai Z, Li J, Dong C, et al. FOXO3-Activated circFGFBP1 Inhibits Extracellular Matrix Degradation and Nucleus Pulposus Cell Death via miR-9-5p/BMP2 Axis in Intervertebral Disc Degeneration In Vivo and In Vitro. Pharmaceuticals (Basel). 2023;16(3).
-
Min Q, Chen X, Yifei G, Baifeng S, Zichuan W, Xiaolong S, et al. FOXO3a overexpression ameliorates intervertebral disc degeneration by decreasing NLRP3-mediated pyroptosis. Int Immunopharmacol. 2025;144:113596.
-
Xu C, Vitone GJ, Inoue K, Ng C, Zhao B. Identification of a Novel Role for FOXO3 Isoform2 in Osteoclastic Inhibition. J Immunol. 2019;203(8):2141-9.
-
Stein CA, Hansen JB, Lai J, Wu S, Voskresenskiy A, Hog A, et al. Efficient gene silencing by delivery of locked nucleic acid antisense oligonucleotides, unassisted by transfection reagents. Nucleic Acids Res. 2010;38(1):e3.
-
Cohen SA, Bar-Am O, Fuoco C, Saar G, Gargioli C, Seliktar D. In vivo restoration of dystrophin expression in mdx mice using intra-muscular and intra-arterial injections of hydrogel microsphere carriers of exon skipping antisense oligonucleotides. Cell Death Dis. 2022;13(9):779.
-
Bennett CF. Therapeutic Antisense Oligonucleotides Are Coming of Age. Annu Rev Med. 2019;70:307-21.
-
Scott AJ, Ellison M, Sinclair DA. The economic value of targeting aging. Nature Aging. 2021;1(7):616-23.
-
Campins L, Serra-Prat M, Palomera E, Bolibar I, Martinez MA, Gallo P. Reduction of pharmaceutical expenditure by a drug appropriateness intervention in polymedicated elderly subjects in Catalonia (Spain). Gac Sanit. 2019;33(2):106-11.
-
Ponnappan RK, Markova DZ, Antonio PJ, Murray HB, Vaccaro AR, Shapiro IM, et al. An organ culture system to model early degenerative changes of the intervertebral disc. Arthritis Res Ther. 2011;13(5):R171.